Abstract
We originally identified T-cell immunoglobulin mucin-3 (TIM-3) as a leukemic stem cells (LSCs)-specific surface molecule and a useful marker for discriminating LSCs from hematopoietic stem cells (HSCs). Furthermore, we recently identified an unique autocrine loop composed of TIM-3 and its ligand galectin-9 (Gal-9). This TIM-3/Gal-9 autocrine loop enhances self-renewal capacity of AML-LSCs and contributes to leukemia progression(Kikushige et al., Cell Stem Cell 2015).
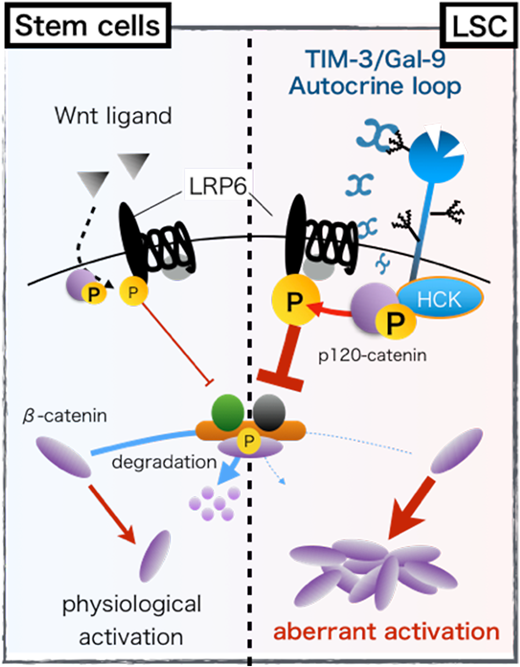
To clarify the molecular mechanism how TIM-3 signaling enhances stem cell properties of AML-LSCs, we performed shRNA-mediated knock-down(KD) of TIM-3 in KASUMI-3 (TIM-3+ AML cell line). As a result, TIM-3-KD significantly reduce the proliferation of KASUMI-3. To investigate how TIM-3-KD attenuated leukemia propagation, we compared gene expression profile of scramble-infected control and TIM-3-KD KASUMI-3 cells. Gene Set Enrichment Analysis (GSEA) revealed TIM-3-KD resulted in the significaltly attenuated expression of HSCs and LSCs-related genes (Eppert et al., Nature Med 2011) and canonical Wnt pathway-related genes (BioCarta). Since canonical Wnt pathway is known to regulate stem cell properties via inducing the nucleus accumulation ofβ-catenin in many types of stem cells including HSCs, we next examined the β-catenin level in TIM-3-KD-KASUMI-3 cells. Array scan analysis revealed that KD of TIM-3 significantly impaired the the nucleus accumulation of β-catenin in KASUMI-3 cells. In various types of cancers, the constitutive activation of β-catenin is one of the most common oncogenic signatures, and it is mainly driven by somatic mutations in the genes related to the canonical Wnt pathway. However, such mutations are rarely detected in AML.We, therefore, hypothesized that TIM-3/Gal-9 autocrine loop signaling might be a novel mutation-independent machinery for the constitutive activation of canonical Wnt pathway in primary AML.
To test this hypothethis, we first evaluated the phosphorylation status of LDL receptor-related protein 6 (LRP6), because LRP6 phosphorylation is a crucial step for canonical Wnt pathway activation. Surprisingly, Gal-9 stimulation induced phosphorylation of LRP6 and subsequent β-catenin accumulation in primary TIM-3+ AML cells even in the absence of Wnt ligand. On the other hand, LRP6 phosphorylation induced by Gal-9 stimulation was canceled by the blocking antibody to TIM-3 (F38-2E2). These results indicate that Gal-9 stimulation should induce canonical Wnt pathway activation via TIM-3 dependent manner in primary AML cells. Since Gal-9 ligation to TIM-3 is known to induce phosphorylation of tyrosine residues of TIM-3 cytoplasmic tail, leading to the recruitment and activation of several Src family kinases (SFKs) for downstream signal transduction. We found that TIM-3/Gal-9 interaction recruited hematopoietic cell kinase (HCK), a member of SFKs, to TIM-3 and activated HCK by phosphorylation of the SH1 domain (Y410 in HCK) in TIM-3+ AML cells. Furthermore, we confirmed a specific HCK inhibitor A-419259 efficiently blocked the LRP6 phosphorylation and subsequent β-catenin accumulation induced by TIM-3/Gal-9 interaction in primary AML cells. These results suggest that the canonical Wnt pathway activation induced by Gal-9 ligation to TIM-3 is mainly mediated by HCK in primary AML cells.
We next focused on p120-catenin as a bridging molecule between HCK activation and LRP6 phosphorylation because, as previously reported, this molecule can be directly activated by SFKs and the activation of this molecule is an initial step for canonical Wnt pathway activation preceding LRP6 phosphorylation. We found that Gal-9 ligation to TIM-3 immediately induced the direct association of HCK and p120-catenin, leading to the activation of p120-catenin in TIM-3+ AML cells. We also confirmed that specific inhibition of HCK completely abrogated the activation of p120-catenin and subsequent canonical Wnt pathway activation in primary TIM-3+ AML cells, indicating that the activation of p120-catenin mediated by HCK is a bridging moleculer machinery for connecting TIM-3 signaling and previously identified canonical Wnt pathway. Since TIM-3 as well as HCK is specifically expressed in LSCs but not in HSCs, we conclude that TIM-3/Gal-9 autocrine loop is a novel Wnt-ligands independent machinery for the constitutive activation of canonical Wnt pathway specific for AML-LSCs. (summarized in figure)
Akashi:Celgene: Research Funding, Speakers Bureau; Pfizer: Research Funding; Bristol-Myers Squibb: Research Funding, Speakers Bureau; Kyowa Hakko Kirin: Research Funding, Speakers Bureau; Taiho Pharmaceutical: Research Funding; MSD: Research Funding; Asahi-kasei: Research Funding; Chugai Pharma: Research Funding; Ono Pharmaceutical: Research Funding; Otsuka Pharmaceutical: Research Funding; Eli Lilly Japan: Research Funding; Novartis pharma: Research Funding; Eisai: Research Funding; sanofi: Research Funding; Astellas Pharma: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal